Research
RESEARCH
We are a systems neuroscience laboratory investigating the neurobiological basis of fear memory, depression, and anxiety. Our research encompasses multiple parallel projects, each contributing to our comprehensive understanding of how the brain processes fear memory and associated stress responses. One of our research avenues delves into basal forebrain neuromodulation of the amygdala. In this pursuit, we integrate viral tract-tracing, optogenetics, chemogenetics, behavioral testing, and network modeling to elucidate the role of ventral pallidum in amygdala-dependent stress responses. Another major line of research focuses on unraveling the antidepressant properties of ketamine. We aim to understand the behavioral pharmacology of ketamine and maximize its therapeutic potential. The third main focus of our lab is to develop novel rodent behavioral paradigms and find ways to measure the rodent affective states in natural, stress-free ways.
Basal Forebrain GABAergic Modulation of Acute and Chronic Fear
The basal forebrain nuclei send cholinergic, glutamatergic, GABAergic and peptidergic projections to several limbic structures that underlie diverse forms of memory and affective processes. For instance, GABAergic projections from the medial septal nucleus of the basal forebrain is required for the emergence of hippocampal theta oscillation as well as hippocampal-dependent learning and memory, such as trace eye-blink conditioning and spatial navigation. These septo-hippocampal neurons, therefore, play a significant role in modulating local network computations of this limbic structure. We postulate that similar basal forebrain GABAergic projections to other limbic structures underlie learning processes run by those structures by contributing to local network activity/oscillations. As such, ventral pallidum innervation of the basolateral amygdala may have a key role in conditioned fear learning and acute fear response, while a potential basal forebrain projection to the bed nucleus of the stria terminalis (BNST) may be required for sustained fear or anxiety.
We use anterograde and retrograde tract-tracing combined with fluorescence immunohistochemistry to examine basal forebrain GABAergic and glutamatergic projections to limbic structures and identify their postsynaptic targets. To determine their functional roles, we selectively activate or inactivate specific basal forebrain neuron subgroups using chemogenetics and optogenetics, and assess behavioral outcomes in various paradigms. In collaboration with Prof. Satish Nair’s team at the University of Missouri, we also work on modeling the effects of different basal forebrain projections on amygdala network oscillations (Tuna et al., 2025).
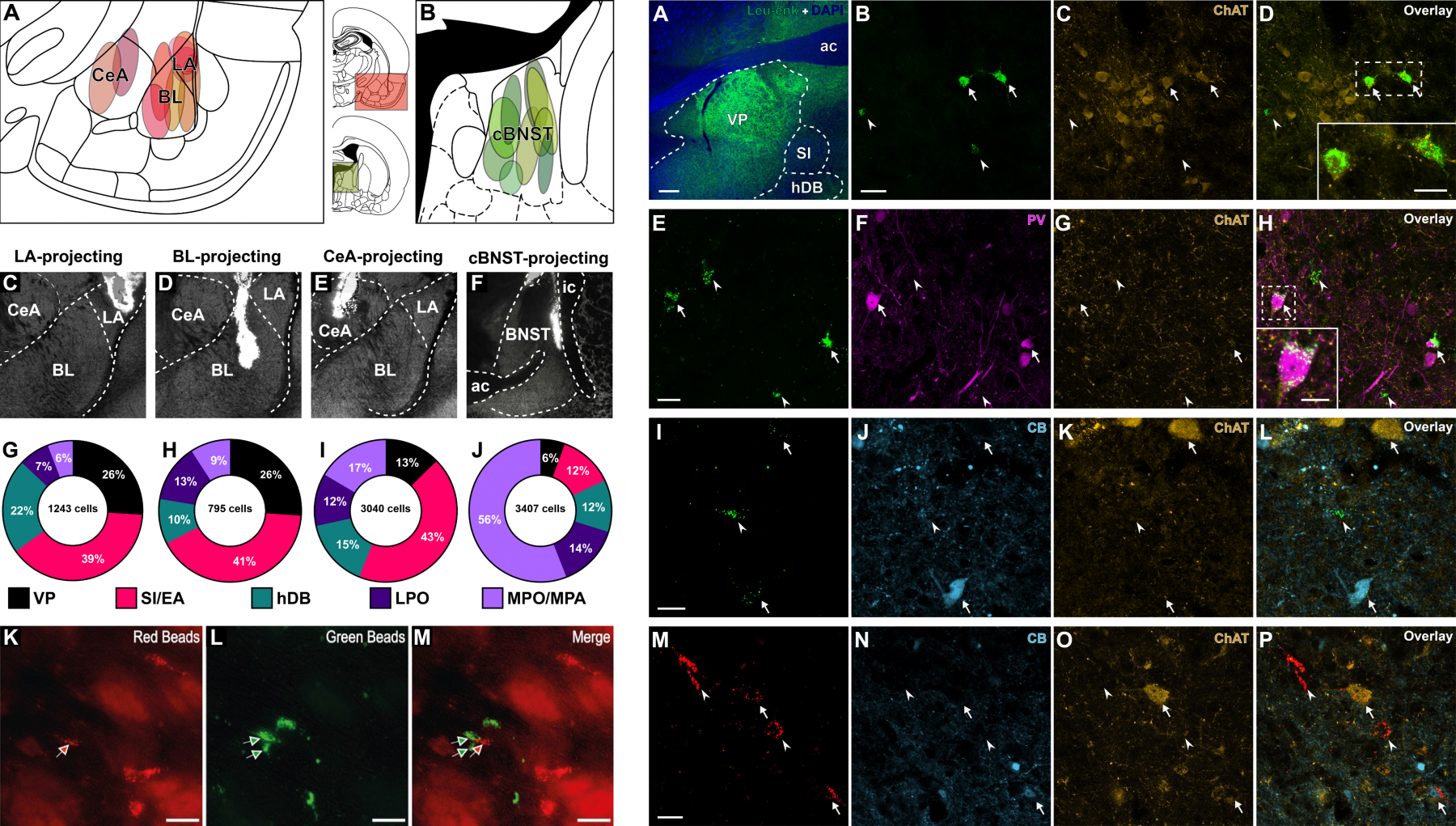
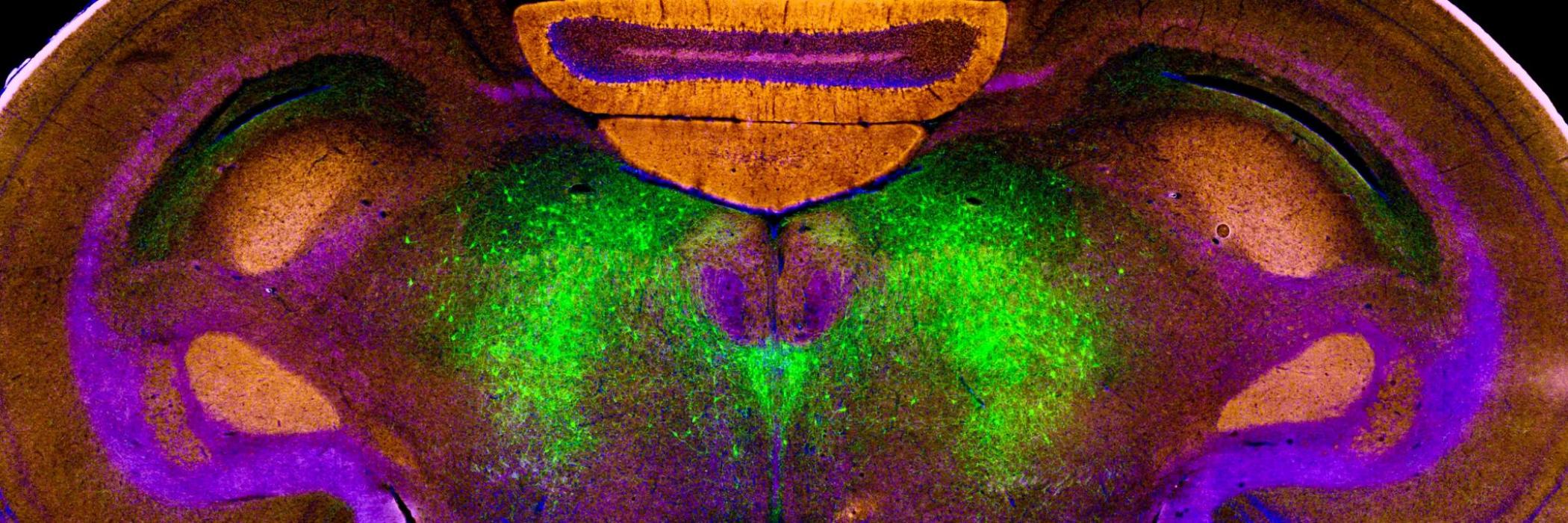
Retrograde tract-tracing of the amygdala and BNST-targeting ventral pallidum (VP), extra-pallidal basal forebrain nuclei and optic area neurons. Figure on the left depicts injection sites, while figure on the right shows immunohistochemical characterization of retrogradely-labeled VP neurons that target different subnuclei of the amygdala and the BNST.
Medial Prefrontal Cortex Innervation of the Basal Forebrain
The medial prefrontal cortex (mPFC) is a key regulator of several limbic functions (including reward-seeking, fear, anxiety, and explicit memory) primarily through its monosynaptic projections to subcortical targets. Its two major subdivisions, the prelimbic (PrL) and infralimbic (IL) cortices, play central roles in integrating affective processes with autonomic and neuroendocrine systems. The PrL sends its densest projections to the nucleus accumbens core, ventral tegmental area, and basolateral amygdala, where it contributes to goal-directed behavior and cognitive control. In contrast, the IL projects most strongly to the nucleus accumbens shell, central amygdala, and the BNST, supporting emotion regulation and fear extinction. Both mPFC regions also send substantial projections to the basal forebrain, the largest neuromodulatory complex in the mammalian brain. We aim to elucidate the specific affective roles of PrL and IL efferents to the basal forebrain within "the cortical-basal forebrain-cortical loop" by using chemogenetic and optogenetic manipulations combined with multi-unit recordings in freely moving rats.
Optogenetic activation of prelimbic and infralimbic neurons projecting to the basal forebrain. The experimental timeline and behavioral test results are presented across baseline, pre-stimulation, stimulation (cyan), and post-stimulation epochs for control (black, inverted triangles), prelimbic activation (magenta, circles), and infralimbic activation (green, squares) groups. Bilateral optic fibers were implanted in the basal forebrain, with fiber tips positioned in the ventral pallidum. Error bars represent SEM. Statistical significance was set at α = 0.05 (*).
Antidepressant Effects of Ketamine
Ketamine, a dissociative non-competitive N-methyl-D-aspartate receptor (NMDAR) antagonist, has mainly been used as a general anesthetic for children. Its antidepressant properties were discovered recently, placing ketamine as a potent atypical antidepressant drug with a great potential. One of the first animal studies investigating the antidepressant effects of ketamine was published in our laboratory, revealing that a single anesthetic dose of ketamine (160 mg/kg, IP) has a prolonged ameliorative effect on behavioral despair in rats (Yilmaz et al., 2002). Pilot clinical studies on ketamine showed that the drug has promising ameliorative effects on a number of depressive symptoms, including suicidal thoughts. With a growing body of both animal studies and clinical trials, this anesthetic substance now emerges as a proper antidepressant, especially for those who cannot benefit from traditional forms of treatment. Nasal sprays of ketamine have now been approved by the FDA for adults with treatment-resistant depression.
We have a three-pronged research approach to ketamine. First, we aim to optimize its antidepressant effects by fine-tuning the dose, administration methods, and potential supra-additive interactions with other antidepressants. In this regard, we have shown that sustained, voluntary administration of low-dose oral ketamine in rats produces therapeutic effects in behavioral despair (Ecevitoglu et al., 2019) and anhedonia (Kingir et al., 2023). Expanding on non-invasive applications, we developed a transdermal ketamine model and observed antidepressant effects using a novel shea butter-based ketamine ointment (Akan et al., 2023).
Second, we investigate ketamine’s complex effects on different memory systems. We examined how an antidepressant dose of ketamine (10 mg/kg, IP) influences the encoding, retrieval, and modulation of fear and spatial memory in adult rats (Yuksel et al., 2024). Additionally, we combined acute ketamine administration with environmental enrichment and found that this combinatorial approach ameliorated spatial memory deficits in the Morris water maze (Aykan et al., 2024).
Finally, we integrate behavioral pharmacology with bioinformatics to explore ketamine’s versatile mechanism of action. Our recent work focuses on the role of metabotropic glutamate receptors (Gokalp & Unal, 2024) and estrogen receptors (Idil et al., 2025) in ketamine’s therapeutic effects.
 Searching for the recent AlphaFold literature at the Library of Celsus (left), and trying to identify the NMDAR binding site of rapastinel on the streets of Cunda, following joint lab meetings with Karaca Lab (İBG) and Uyar Lab (İYTE).
Searching for the recent AlphaFold literature at the Library of Celsus (left), and trying to identify the NMDAR binding site of rapastinel on the streets of Cunda, following joint lab meetings with Karaca Lab (İBG) and Uyar Lab (İYTE).
Novel Methods to Assess and Manipulate the Affective States of Rodents
One of our ongoing efforts focuses on employing more naturalistic methods to assess the affective states of rodents in behavioral testing. Traditional animal tests typically evaluate affective behaviors by exposing rodents to mildly to moderately aversive conditions, thereby motivating them to respond differently based on their affective state. Common examples include measuring behavioral despair in the forced swim test or assessing anxiety-related behaviors in the elevated plus maze. To develop more ecologically valid approaches, we focus on methods such as recording microexpressions and ultrasonic vocalizations (USVs), replacing Skinner boxes and experimental chambers with mobile and interactive robots (Nas et al., 2025), and designing novel behavioral models and tests that incorporate naturalistic elements (Gencturk & Unal, 2024).
USVs provide key insight into the affective states of rodents. The typical human hearing range falls within 20 Hz to 20 kHz, limiting our ability to perceive sounds beyond this spectrum. In contrast, rodents possess a remarkable capacity to both emit and detect sounds at frequencies exceeding 20 kHz, which are therefore designated as ultrasonic. These vocalizations serve as a crucial mode of intraspecies communication for rodents and offer insights into their acute emotional states. Rat pups emit stress-induced ultrasonic vocalizations, particularly during separation from their mothers (see the figure below). In contrast, adult rats emit 50 kHz vocalizations when displaying positive, appetitive behavior; while bursts of 22 kHz USVs indicate aversive or distressing states. The latter function as alarm calls in response to perceived danger, such as the presence of a natural predator. Intriguingly, these 22 kHz vocalizations have been suggested to be the rodent equivalent of human crying. In our laboratory, we employ recording and playback systems capable of capturing and producing frequencies up to 120 kHz. Our primary objective is twofold: to enhance our ability to monitor the emotional states of rats and to gain a deeper understanding of the nature and significance of these vocalizations. In addition, we harness artificial USVs to manipulate rodent behavior. For instance, a series of 22 kHz pulses generated from an artificial source can be used to reduce mobility in rats, while 50 kHz emissions can prompt approach behavior.
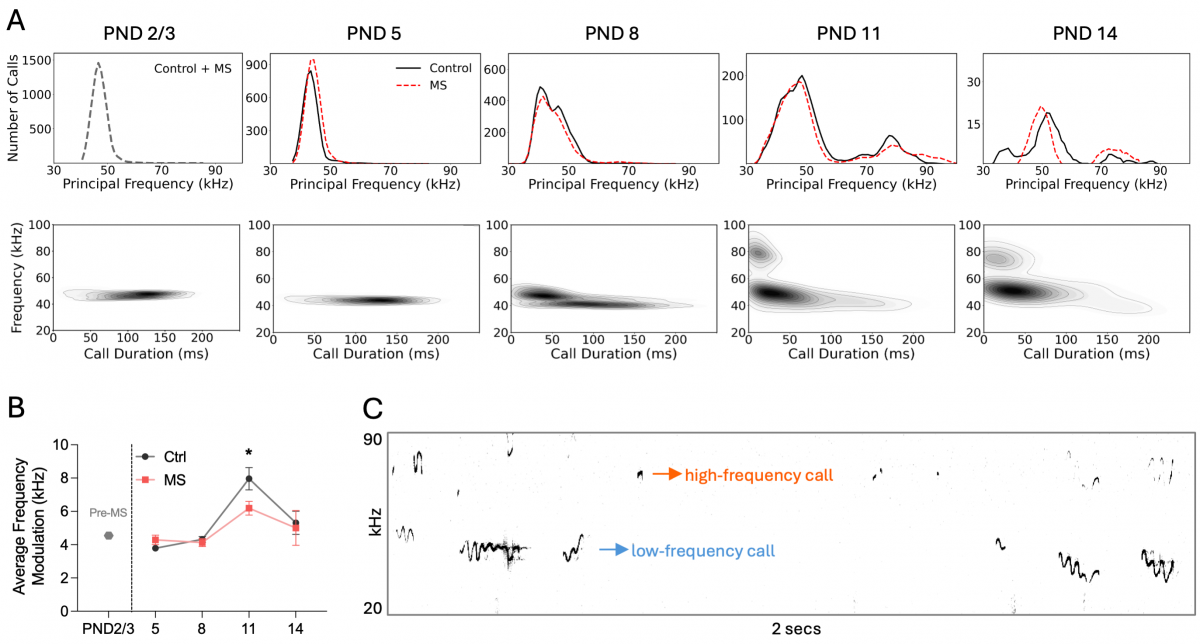
Frequency-related properties and classification of pup USVs. (A) Distribution of calls from control (black) and maternal separation (red) groups across days based on principal frequency (top) and corresponding heatmaps (bottom). (B) Line graph showing average frequency changes in calls across days. (C) Example spectrogram from postnatal day (PND) 11, illustrating low-frequency (<60 kHz, blue) and high-frequency (≥60 kHz, orange) USVs.
As part of these efforts, we participate in the COST Action Affect-Evo: An evolutionary view to understanding affective states across species (https://affect-evo.eu/). This multidisciplinary consortium brings together researchers from the natural and social sciences, philosophy, psychology, animal welfare, and technology, promoting collaboration across Europe to deepen our scientific understanding of affective states and address societal challenges through research networking and capacity building. Our PI leads the Working Group on improving treatments for affective problems across species. This group aims to advance the understanding and treatment of affective disorders—such as depression, anxiety, and pain-related affective components—across different species. Its focus is on translating research findings into practical interventions for both animal models used in biomedical research and animals receiving direct care, including companion and zoo animals. Expected outcomes include best-practice recommendations for diagnosing and managing affective disorders, contributing to improved animal welfare and more robust human–animal comparative models.
The Limbic System of the Japanese Quail
Until the early 2000s, the prevailing belief was that the avian brain was primarily composed of the striatum. However, a more contemporary perspective has emerged, identifying the pallium and subpallium as analogous to the mammalian cortex and subcortex, respectively (Reiner et al., 2004; Jarvis et al., 2005). This revised understanding of the avian brain has spurred a growing interest in avian research, particularly in the realm of cognitive processes. Japanese quails, in particular, have proven to be ideal avian subjects for investigating appetitive conditioning.
In collaboration with Prof. Onur Güntürkün’s team at the Ruhr University Bochum, we analyze and characterize brain structures responsible for cognitive and affective processing in quails. We employ a diverse range of techniques, including histological staining, immunohistochemistry (IHC), immunofluorescence (IF), and the iDISCO tissue clearing method. These methods enable us to distinguish and visualize distinct brain regions and the major neuromodulatory pathways, such as the dopaminergic and noradrenergic systems, in the quail brain. We also use a combination of Golgi-Cox staining and immunofluorescence to observe the intricate morphological features of neurons that have been neurochemically identified within the limbic structures. Additionally, we conduct fiber density analysis of dopaminergic fibers and investigate their basket-like axonal formations. This analysis helps us differentiate the nidopallium caudolaterale (NCL), which corresponds to the mammalian prefrontal cortex in quails.
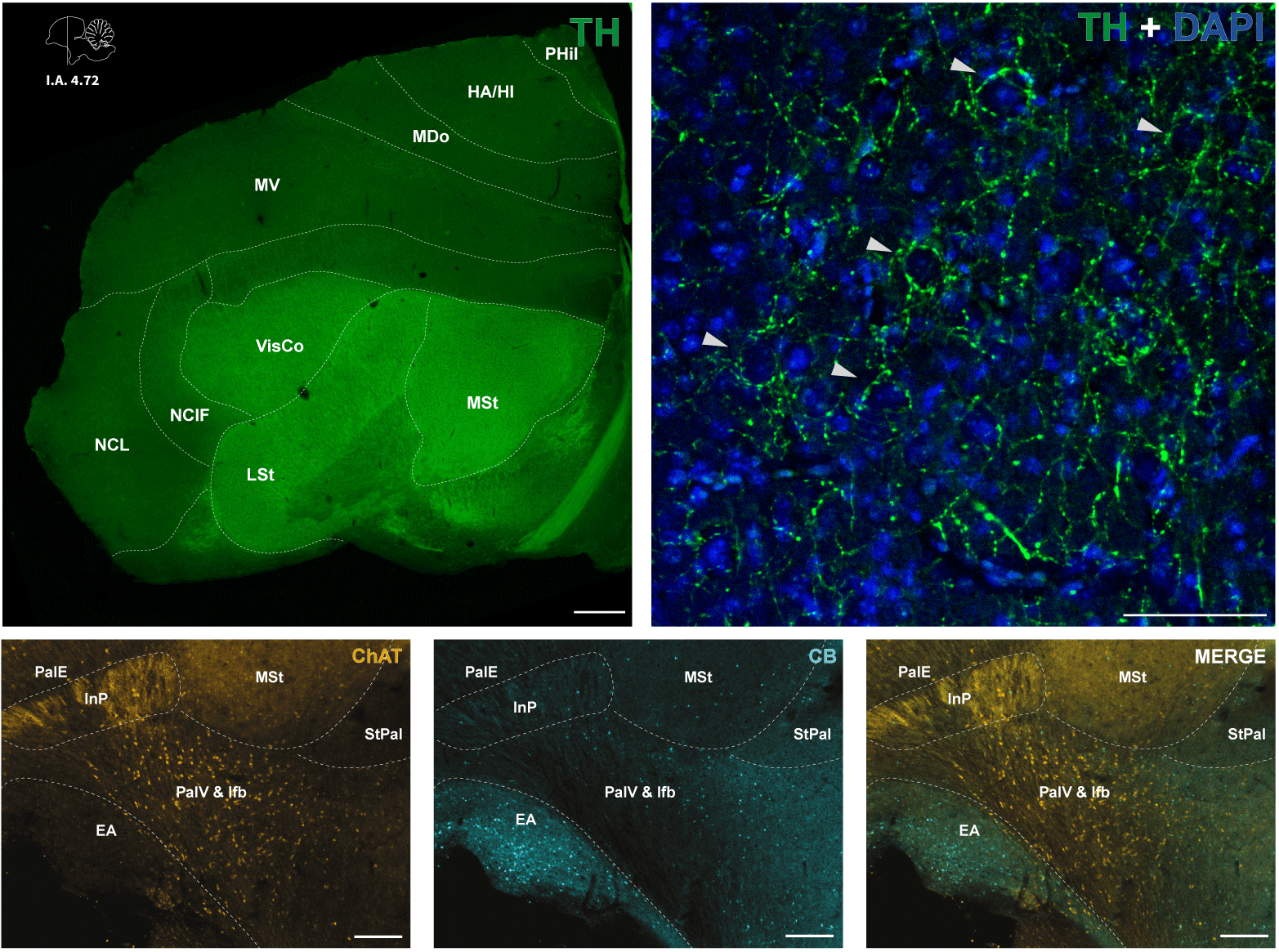 Coronal section of a single hemisphere of the Japanese quail brain depicting differentiation of the nidopallium and striatal regions with tyrosine hydroxylase (TH) immunopositive processes (upper left). TH-positive axons form characteristic basket-like formations (arrowheads) around the somata of DAPI-stained nuclei (upper right). The expression of calbindin (CB) and choline acetyltransferase (ChAT) immunopositive cells and fibers in limbic regions (bottom). Scale bars: 50 μm.
Coronal section of a single hemisphere of the Japanese quail brain depicting differentiation of the nidopallium and striatal regions with tyrosine hydroxylase (TH) immunopositive processes (upper left). TH-positive axons form characteristic basket-like formations (arrowheads) around the somata of DAPI-stained nuclei (upper right). The expression of calbindin (CB) and choline acetyltransferase (ChAT) immunopositive cells and fibers in limbic regions (bottom). Scale bars: 50 μm.